The South African government’s decision to procure pneumococcal vaccines from India-based Cipla, rather than from local manufacturer Biovac (a bio-pharmaceutical company partly owned by the South African government), came hot on the heels of the World Health Organization’s (WHO’s) launch of an mRNA technology transfer hub at Afrigen Biologics in Cape Town.
The move to switch to a cheaper supplier highlights tensions between price pressure (a saving of roughly R2.4-billion over three years), local production and sovereignty.
In the broader context of continent-wide efforts to increase Africa’s vaccine manufacturing capabilities, the decision highlights the fundamental importance of having a harmonised approach to procurement as part of a coherent strategy that enables local manufacturing.
The stakes are high and deeply consequential for Africa’s development.
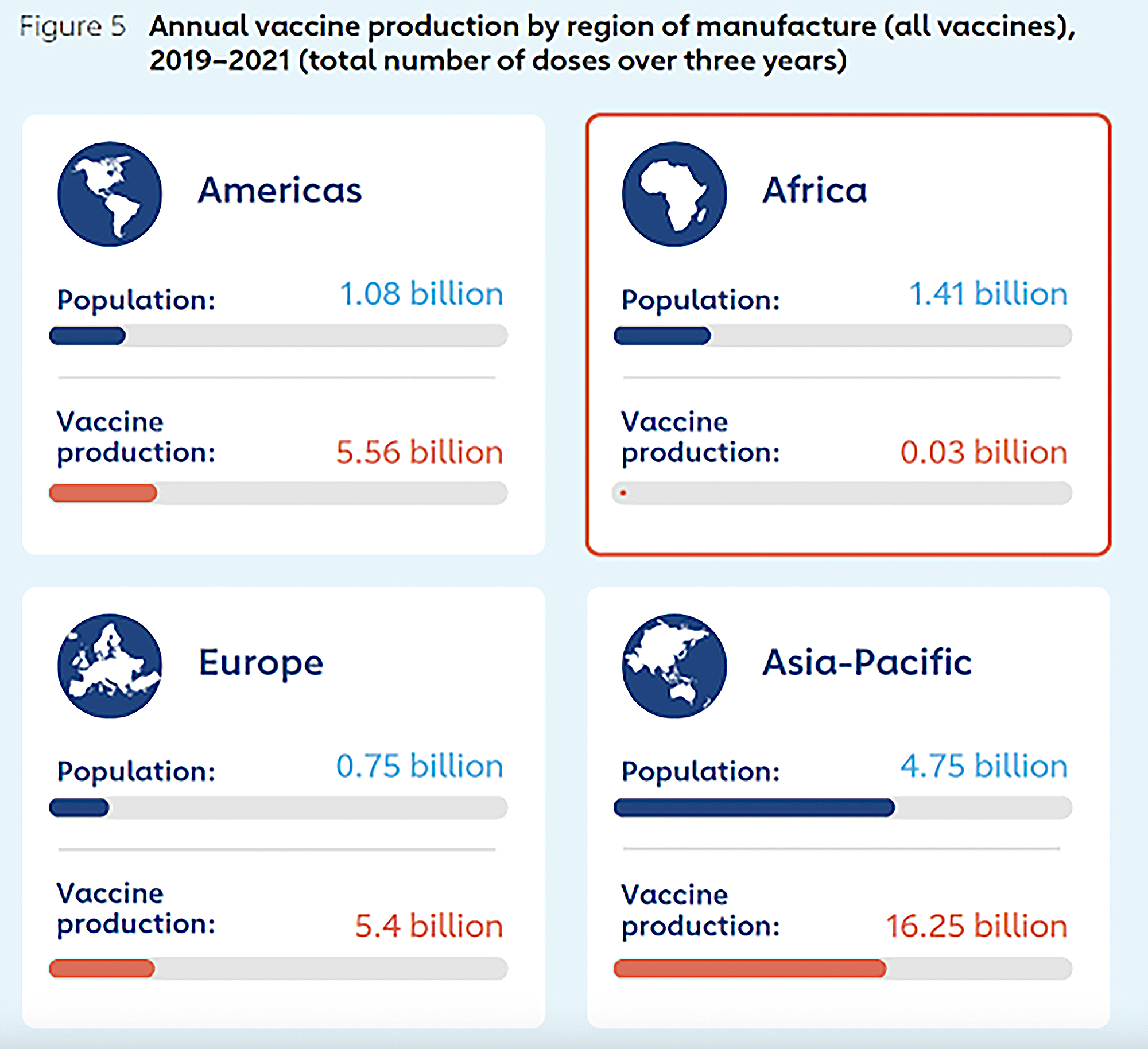
The continent is currently the only major world region that does not have any significant domestic vaccine manufacturing capacity. This has led to a long-standing cycle of dependence on foreign aid and procurement of vaccines that has hindered any efforts to build large-scale manufacturing capabilities.
The devastating consequences of this dynamic were seen most recently during the Covid pandemic: delays in procurement and distribution of both routine vaccines and Covid vaccines led to a reversal in decades of hard-won routine vaccine coverage and, as of 27 April 2023, only 30.8% of the African population has been fully vaccinated against Covid.
An important rallying point for developing vaccine manufacturing in South Africa and on the continent is the Partnerships for African Vaccine Manufacturing (PAVM), established by the African Union in 2021.
The PAVM Framework for Action sets the goal of producing 60% of Africa’s vaccines on the continent by 2040. Acknowledged in the framework is the need for agreements from African countries to initially pay a premium for locally produced vaccines in order to support nascent manufacturers.
South Africa’s decision to do exactly the opposite threatens to hinder the goals set forth by the PAVM, thereby setting an unfortunate precedent, and ultimately reflecting an uncoordinated vaccine procurement policy.
The largest purchaser of vaccines for Africa is currently the Global Alliance for Vaccines and Immunization (Gavi). About 90% of Africa’s vaccine procurement volume is driven by Gavi, with self-financing countries accounting for the remaining 10%.
Strikingly, Gavi-supported countries pay about one-third of the price-per-dose compared with their self-financing peers.
This unsustainable dynamic is currently one of the largest hindrances to new manufacturers on the continent, as nascent vaccine firms will initially have to charge significantly more per dose to offset training, start-up and operation costs.
As the largest purchaser in the African vaccine market, Gavi has recently committed to supporting the PAVM through its 10-point action plan. In addition to providing guidelines for its philanthropic and government partners to support African vaccine manufacturers, Gavi has suggested that it would support advance market commitments by leveraging its role as a market shaper.
If South Africa’s government can develop a more coherent policy surrounding the manufacture and procurement of vaccines, which will require partnering successfully with the private sector, the economic dividends and healthcare benefits of the resulting biotech and vaccine manufacturing ecosystem will be substantial.
Ironically, India can serve as a case study.
In 1986, India became one of the first countries to have a government unit dedicated to biotech with a budget of $835,000. After three decades of concerted effort, favourable regulations and investment in human capital, India’s biotech industry created hundreds of thousands of direct and indirect jobs and reached a valuation of $70.2-billion in 2020. It is expected to be valued at $150-billion by 2025. South Africa can and must embark on a similar journey.
Moving forward, South Africa can provide greater support for local manufacturers by restructuring its vaccine manufacturing and procurement policy to include advance market commitments, investing in human capital to support the vaccine manufacturing industry, providing a streamlined and efficient vaccine regulatory environment, and providing greater support for the country’s research and development infrastructure.
(Greater detail on such a strategy can be found in a report prepared for the South African government titled, South African Vaccine Manufacturing Capabilities: Key Findings and Capabilities, which will be released on 4 May during a webinar on vaccine manufacturing in Africa).
Pursuing such a strategic approach will require significant upfront investment, but South Africa has a pre-existing infrastructure that can be leveraged:
- South African vaccine manufacturers such as Biovac already possess significant fill-and-finish capacity and are already in the process of upscaling their end-to-end manufacturing capabilities.
- Similarly, Aspen Pharmacare Holdings, who, during the Covid-19 pandemic, had repurposed one of their manufacturing plants to allow them to fill and finish vaccines, has recently concluded a 10-year agreement with the Serum Institute allowing Aspen to manufacture four Aspen-branded vaccines, from bulk drugs supplied by the Serum Institute of India, for marketing and distribution to selected organisations and markets in Africa.
- Beyond this, and in line with the One-Health concept (that recognises the interconnection between people, animals and plants and their shared environment), Onderstepoort Biological Products, a South African government-owned company, is well-established as a manufacturer of vaccines for the veterinary market.
There is also no shortage of South African universities capable of supporting robust research and development programmes and providing high-level talent, while the South African Medical Research Council and National Institute for Communicable Diseases of South Africa are capable of further supporting research efforts and helping to train a resilient workforce. From the regulatory perspective, the South African Health Products Regulatory Authority has a well-earned reputation as an exacting and rigorous medicines approval agency.
With the recent launch of the WHO’s mRNA hub at Afrigen, South Africa has the potential to become a continental leader in vaccine development and manufacturing.
Ultimately, the government’s decision to procure vaccines from Cipla, because of the absence of a coordinated interministerial decision-making capacity, is a short-sighted one that undermines ongoing efforts in South Africa, and on the continent, to develop domestic vaccine manufacturing.
To rectify the situation, the South African government must reconsider its decision and show much greater support for local manufacturing.
It is the right thing to do for the country’s people and industry, and is also critical in supporting continent-wide efforts to achieve vaccine independence.
As the recent pandemic has shown, R2.4-billion is a modest price to pay to ensure that this happens. DM
Dr Wilmot James is a Professor (Health Services, Policy and Practice) at the School of Public Health and Senior Adviser to the Pandemic Center at Brown University, Rhode Island. Professor Martin Veller is the former dean of the Faculty of Health Science at the University of the Witwatersrand. They both thank Lewis Rubin-Thompson from the University of Rochester for his assistance.




 Vaccines exit a vial monitor-labelling machine at the Serum Institute of India's pharmaceutical plant in Pune, Maharashtra, India, on 4 May 2015. (Photo: Sanjit Das / Bloomberg via Getty Images)
Vaccines exit a vial monitor-labelling machine at the Serum Institute of India's pharmaceutical plant in Pune, Maharashtra, India, on 4 May 2015. (Photo: Sanjit Das / Bloomberg via Getty Images)