The latest estimates from Thembisa, the leading mathematical model of HIV in South Africa, show that the rate of new HIV infections in South Africa is declining, but at 164,000 in 2022, it is still very high.
Rather than a single silver bullet, several delegates at the conference called for a basket of HIV prevention options suited to people’s (especially young women’s) needs.
/file/dailymaverick/wp-content/uploads/2023/06/pimart1.jpeg)
The most recent option to add to this basket is for people who are not living with HIV to take specific antiretrovirals to prevent HIV infection. Such pre-exposure prophylaxis (PrEP) can come in the form of a pill taken daily, a monthly vaginal ring, or an injection administered every two months.
More established means of HIV prevention include condoms, voluntary medical male circumcision, and antiretroviral treatment – the latter is an important form of prevention since most people living with HIV become non-infectious on treatment.
However, it has not been smooth sailing for the new kids on the block in South Africa, particularly the vaginal ring and the injection.
Where we are with PrEP
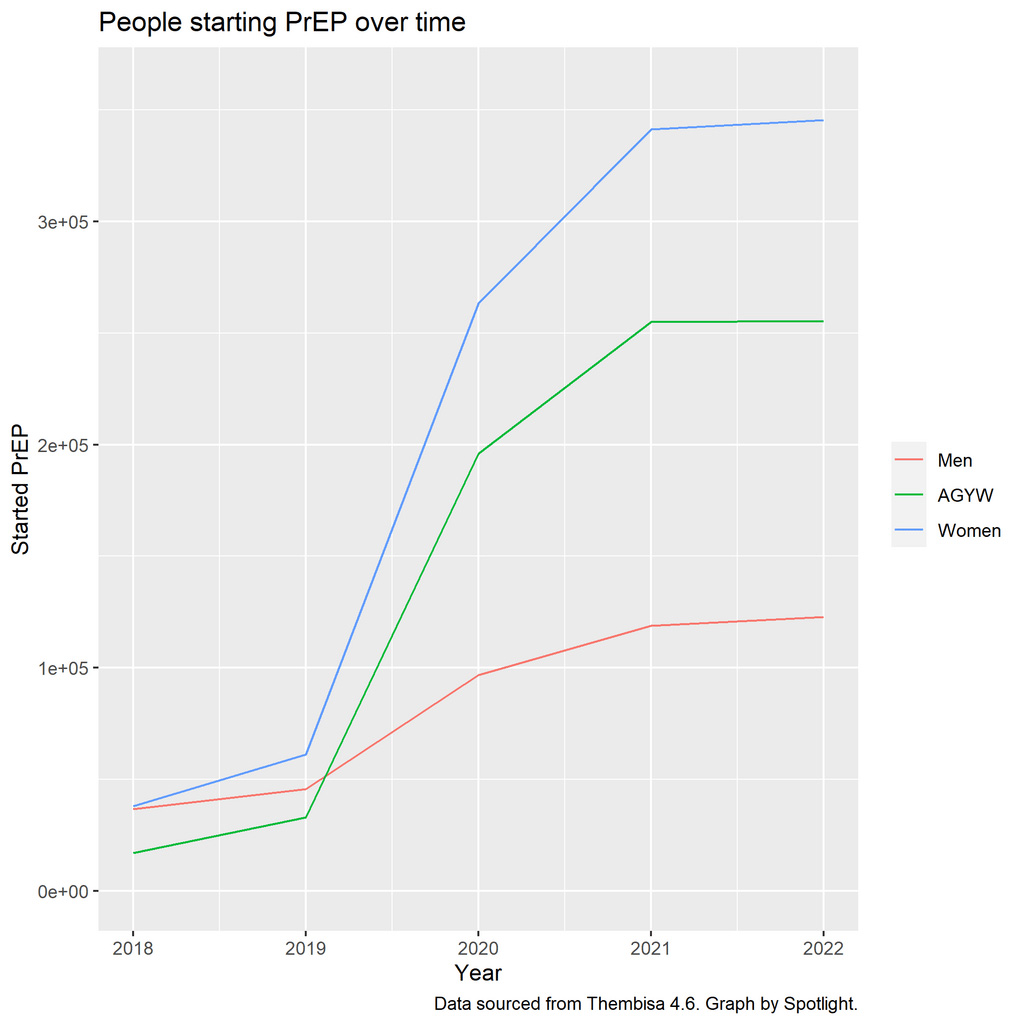
Thus far, oral PrEP, which was approved by the South African Health Products Regulatory Authority (Sahpra) in 2015, has been rolled out in over 2,000 public healthcare sites across the country at the last count in November 2022. Uptake has, however, been slow.
According to Thembisa, only 0.8% of sexually active people in South Africa were taking oral PrEP in 2022 – up from 0.1% in 2018.
Sahpra approved the dapivirine vaginal ring in March 2022. The ring is inserted monthly and is indicated for women 18 years and older. It has been shown to decrease the risk of contracting HIV by around 30% in clinical trials, although there is some reason to think real-world efficacy may be higher.
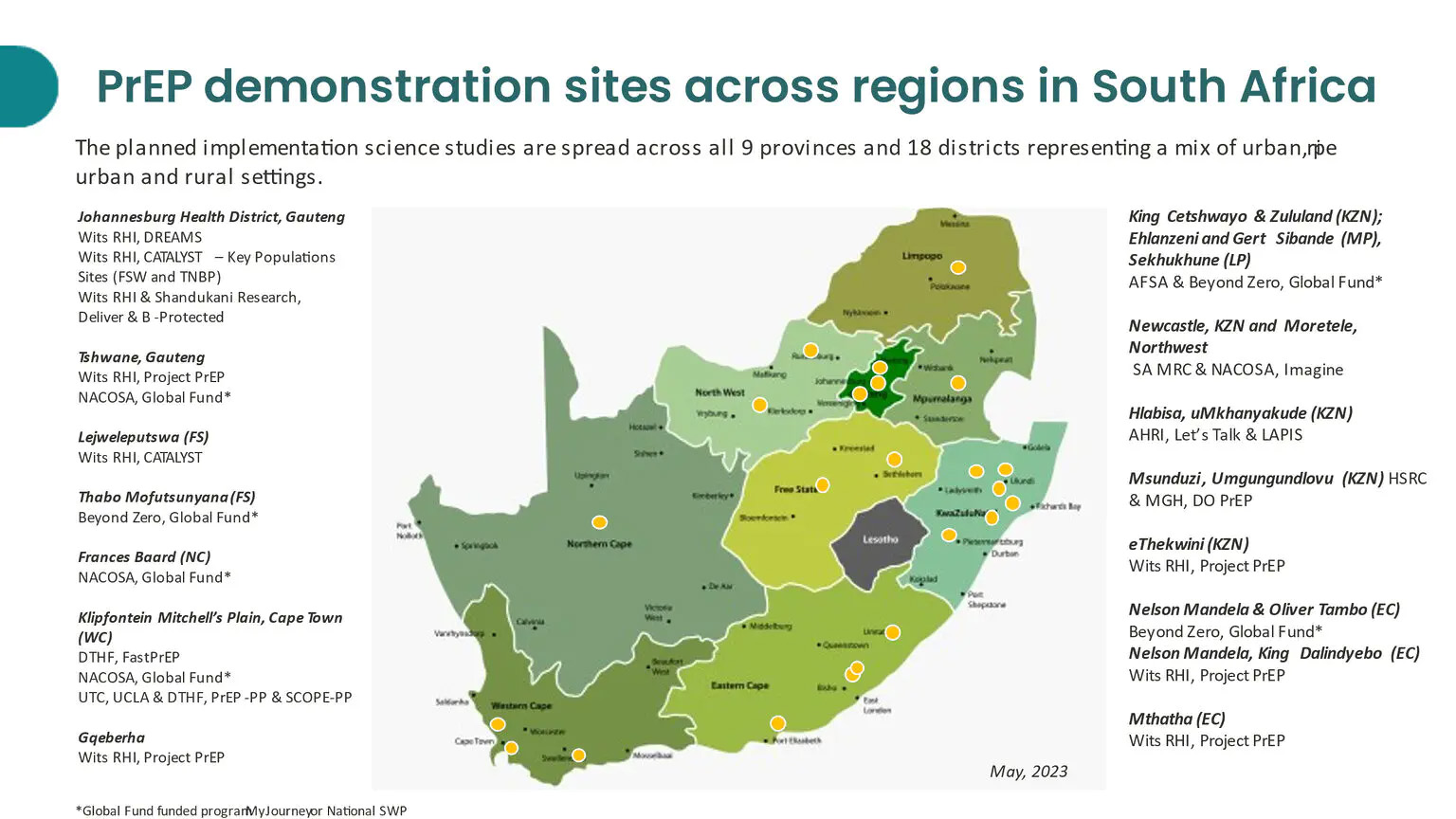
Implementation studies are planned to determine the feasibility of the ring in the South African context. Nicolette Naidoo, the technical head of research for implementation science at the Wits Reproductive Health and HIV Institute (Wits RHI), told Spotlight at the SA Aids conference the studies are only expected to start on 1 July this year.
In December last year, Sahpra authorised the use of the long-acting HIV prevention injection containing the antiretroviral medication cabotegravir (CAB-LA) for use in the country.
The CAB-LA injection administered every two months has been shown to be extremely effective in two large clinical trials. The high efficacy of the injection in these trials, compared to pills or the ring, is thought to be largely driven by the higher levels of treatment adherence associated with the injection.
But how good adherence will be outside of a study context is not known.
To roll out the injection, as with the vaginal ring, implementation studies or pilot projects are needed to assess how these products will perform in the South African context. Spotlight previously reported that CAB-LA pilots were expected to start early in 2023.
/file/dailymaverick/wp-content/uploads/2023/06/Image-21vaccinetrial.jpg)
Dr Sandile Buthulezi, director-general of the National Department of Health, told Spotlight at the conference there have been delays and that a technical working group, comprising clinical experts drawn from various research projects and policy experts, are still finalising protocols.
Why these pilots are important
“There are a number of PrEP introduction studies that are planned in the coming year to introduce cabotegravir and the dapivirine ring in South Africa,” Prof Sinead Delany-Moretlwe, the director of research at Wits RHI, told delegates in a plenary at the SA Aids conference.
These studies will be important, according to Delany-Moretlwe, because they will show how to deliver CAB-LA and the ring in the context of the South African health system; how to deliver differentiated and simplified services for a range of populations; how to deliver HIV testing as well as reveal more information on the cost and benefits for HIV infections averted, and what the demand for the products will be.
There are still some key questions around CAB-LA that need to be answered in a study setting, Naidoo said, like the potential for HIV drug resistance and what the most sensitive testing algorithms would be for detecting HIV infection.
“But also, it is an intramuscular injection, so the questions around feasibility are also really important. Does it [the injection] need to only be done in a facility? Can it be done in a community? Through these studies, they will be answering really critical questions to inform the scale-up of these products,” Naidoo said. (Spotlight previously reported on the importance of these pilots here.)
CAB-LA delays
Responding to follow-up questions on the CAB-LA pilots, national health spokesperson, Foster Mohale, said the pilot studies for CAB-LA are awaiting donations from ViiV, the manufacturer of the drug.
“The delay has been due to challenges with the packaging of the donated product not meeting the South African regulatory requirements,” he said.
Explaining this, he said when Sahpra approves a product, they make certain specifications around packaging requirements for use in South Africa and every product that comes into the country has to be packaged for use here.
Since the donated product does not meet these specifications, this can mean that the originally intended pilots as implementation science studies, which are real-life study environments, may have to be changed to 3B studies, which is more like a clinical trial and different to what was initially envisaged.
He said that ideally, the department prefers it to be a study which is closest to real life and where they can actually offer oral PrEP, with the dapivirine ring, and CAB-LA together so that people can have a choice across these products.
Mohale stressed that the delay is not caused by the development of the protocols and training materials.
“These are already in draft form and they are currently being reviewed and the department will ensure that these are available to guide implementation.” He said clinical guidelines and protocols are required to guide healthcare workers on how to initiate a person on any new medication.
“Professional nurses in PHC (primary healthcare) facilities will be providing CAB-LA. These nurses,” he said, “assess, diagnose, prescribe medications, and provide ongoing management using standardised protocols, job aids, data collection tools, training, and IEC materials.
“So these protocols and job aids provide standardised guidance to nurses to safely administer CAB-LA, especially with regard to, among others, assessments and tests required prior to initiating a person on CAB-LA, dosage and administration of the medication, and monitoring of HIV status, side effects, and any other problems that may result from the use of CAB-LA.”
When asked about the delays in starting the CAB-LA studies, Naidoo said, “I even wouldn’t say it's a delay, because essentially the product is not in the country yet. [The] government (Sahpra) approved the product. We’ve taken the critical step. The next step in our supply chain for a new product is obviously then making sure it gets into the country. And I think that is where we're probably maybe one or two steps behind the DVR [dapivirine ring], but essentially, you are going to see studies launched this year with CAB-LA.”
/file/dailymaverick/wp-content/uploads/2023/06/Linda-Gail-BekkerNew-1024x768-1.jpg)
Also responding to questions relating to the CAB-LA pilots, Prof Linda-Gail Bekker, Desmond Tutu Health Foundation CEO and director of the Desmond Tutu HIV Centre at the University of Cape Town acknowledged that the South African pilots that will be using CAB-LA “hit a bit of a roadblock on getting the product into South Africa because we still need to get it labelled for South Africa”.
“ViiV is making available enough CAB-LA for the pilots so each of us (research organisations involved in the pilots) have had to say how much we need and all that’s gone through and approved,” she said.
The Foundation and Wits RHI are among the research entities involved in the pilots.
According to Bekker, the pilots were expected to start around July this year. “But it’s probably more like September now.”
Cost concerns
The other spanner in the works is the price of both CAB-LA and the dapivirine ring.
Mohale said the national essential medicines list committee considered both the dapivirine ring and CAB-LA as pre-exposure prophylaxis interventions. When this committee considers whether something needs to be included on the national essential medicines list, it looks at various things, including which products are available, as well as cost.
“Because of the costs of the dapivirine ring – and the costs are quite astronomical, they weighed it against oral PrEP, which is costing about R52 for a month’s supply, whereas the dapivirine ring costs in the region of over R300,” Mohale explained.
So, for the dapivirine ring, the committee did not recommend inclusion on the list, but they will reconsider this once the price is comparable to oral PrEP.
With CAB-LA, however, the department still does not have an absolute price at which we would get CAB-LA, he said.
“Right now we’re still talking about the donated product, but the actual price at which we're going to get it, we don’t quite know.”
According to him, a price between £32 and £35 for a two-month dose for lower- and middle-income countries was talked about.
“But they were still going to come up with an absolute price and we don't have an absolute price.”
Bekker also told Spotlight they are waiting to see what the final price point is “in terms of affordability and Treasury being able to bring product in”.
Another variable at play is the limited global supply of CAB-LA, with only one company that makes CAB-LA – ViiV.
“The Medicines Patent Pool got involved now so there will be an opportunity for generic companies to at least fill-and-finish or even start manufacturing, but there’s going to be a lead time to that,” Bekker said.
“So yes, it does look like we do have some delays ahead of us. Hopefully, the pilots will start soon and we will at least be able to learn some aspects. It is worth knowing that the treatment injectables are already being used in a number of implementation science projects and at least we are learning valuable information in that sense. So we should keep optimistic,” she said.
A representative from the Medicines Patent Pool last year told Spotlight that it is expected to take three to five years before generic versions of CAB-LA will become available.
ViiV responds
A ViiV spokesperson this week told Spotlight the company had committed to offer a not-for-profit price for public programmes in low-income, least developed, and all sub-Saharan African countries until a generic is available, including South Africa.
“We are also working strategically with major global health partners who have a shared ambition to enable access to cabotegravir LA for PrEP. Additionally, we recently announced progress on our new voluntary licence with Medicines Patent Pool to help enable access as MPP signed sub-licenses with Aurobindo, Cipla, and Viatris to produce generic versions of cabotegravir LA for PrEP. Both ViiV and MPP will be working closely with these partners to enable generic product development, manufacturing, and supply of cabotegravir LA for PrEP as quickly as possible.
“We were pleased to receive South African regulatory approval, including approved packaging, for cabotegravir LA for PrEP and we are ambitious for the impact that we can have together with global partners to enable access to this new HIV prevention option,” the ViiV spokesperson told Spotlight.
“To help inform public health policies and practices, we have committed supply and support for multiple studies across multiple geographies, including implementation science studies in South Africa. We will continue to work with study sponsors as they work through local regulatory processes to help enable initiation as quickly as possible.”
Regarding supply, ViiV’s spokesperson said, “We are committed to scaling up the manufacture of cabotegravir LA for use in treatment and prevention. At the current time, and while we build up the manufacturing capacity, supply will be balanced across global treatment and prevention programmes, as well as supporting multiple implementation science studies, post-trial access commitments, and some programmatic cabotegravir LA for PrEP roll-out.”
Dapivirine ring projects ready to start
Meanwhile, according to Mohale, the dapivirine ring pilots were due to start in June but there was an unanticipated delay for the post-importation testing that needed to happen.
“But we are now ready to start in July. We have a guideline. We have job aids. We have trained all the people that are going to be implementing this and they are all due to start and as we speak right now, the dapivirine rings that were donated are being distributed to the study sites.”
He said the department has already received a donation of 38,400 dapivirine vaginal rings from USAID to use in implementation science pilot projects that have approved ethical and IRB approval. The department has identified approximately 10 such projects that meet the requirements to utilise these donated dapivirine rings.
“The dapivirine ring implementation guidelines with the job aids, recording and reporting tools for healthcare workers, and information materials for healthcare users were developed and approved in December 2022. Training of all the implementers was conducted in May 2023,” he said.
“The donated dapivirine rings arrived in South Africa in May 2023. These projects are located in all nine provinces.”
Naidoo said at the study sites, the ring will be offered alongside oral PrEP. To access the ring, people will have to enrol in one of the studies, which will provide useful data like what the preference is for the ring and whether participants will switch between the ring and oral PrEP.
“They are implementation science studies, so they will be happening in local clinics and real-world settings in communities and will answer the key questions regarding acceptability, feasibility and so on,” Naidoo said.
She also said that data from the studies will be shared regularly with the National Department of Health.
“The National Department of Health has a monitoring framework that will be collecting data from the implementation science studies. So, data really will be informing the roll-out timeously, not at the end after two or three years,” she said. DM
This article was published by Spotlight – health journalism in the public interest.





 Dapivirine vaginal ring and tablets of oral PrEP medication. (Photo: NIH Image Gallery)
Dapivirine vaginal ring and tablets of oral PrEP medication. (Photo: NIH Image Gallery)