The South African Health Products Regulatory Authority (Sahpra) has issued a warning for members of the public not to use four baby cough medicines that they say might have been brought into the country.
These medicines are not registered in South Africa and according to the World Health Organization they contain high levels of chemicals that can damage children’s kidneys.
Fears are that these are being distributed at informal markets.
The four products are Promethazine Oral Solution, Kofexmalin Baby Cough Syrup, Makoff Baby Cough Syrup and Magrip N Cold Syrup manufactured by Maiden Pharmaceuticals.
The Indian company has not yet issued a response to the order by the World Health Organization but has posted the recall notices on its website.
According to the recall notices issued for these medicines, symptoms of toxic exposure may include headache, vomiting, diarrhoea and the inability to pass urine.
According to Sahpra, all four products contain unacceptable amounts of diethylene glycol and ethylene glycol which are toxic substances and can prove to be fatal.
Visit Daily Maverick's home page for more news, analysis and investigations
“The fact that these medicines were administered to children makes the matter far more serious. Toxic effects following consumption of the product(s) can include abdominal pain, vomiting, diarrhoea, inability to pass urine, headache, altered mental state, and acute kidney injury which may lead to death,” the Sahpra notice reads.
The first deaths were noted in July. Sahpra puts the death toll at 33 of children in Gambia who died of kidney-related illness. But Reuters said in their latest update that the deaths of 66 children are now linked to the medicine.
The WHO conducted laboratory tests which linked the medication to those deaths. The WHO confirmed that the syrups contain diethylene alcohol and ethylene glycol. Both are industrial solvents and harmful.
The Gambia Medicine Control Regulator also issued an order that all products from Maiden Pharmaceuticals must be quarantined.
“Sahpra’s mandate is to ensure that the public has access to quality, safe and effective health products. The safety of the public is of paramount importance. Our Regulatory Compliance unit will work with law enforcement agencies to eradicate any substandard and falsified health products,” Sahpra CEO, Dr Boitumelo Semete-Makokotlela said.
In his remarks on the matter during the weekly WHO press briefing, the organisation’s director Tedros Gebreyesus said the “loss of these young lives is beyond heartbreaking for their families”
“The four medicines are cough and cold syrups produced by Maiden Pharmaceuticals Limited, in India. WHO is conducting further investigation with the company and regulatory authorities in India. While the contaminated products have so far only been detected in Gambia, may have been distributed to other countries. WHO recommends all countries detect and remove these products from circulation to prevent further harm to patients,” Gebreyesus added. DM/MC
If you are in possession or come across any of these products, please ensure that you do not consume them, nor administer them to children. If you know of such instances, please contact Sahpra’s Regulatory Compliance unit at: Mokgadi.Fafudi@sahpra.org.za or on 012 015 5434





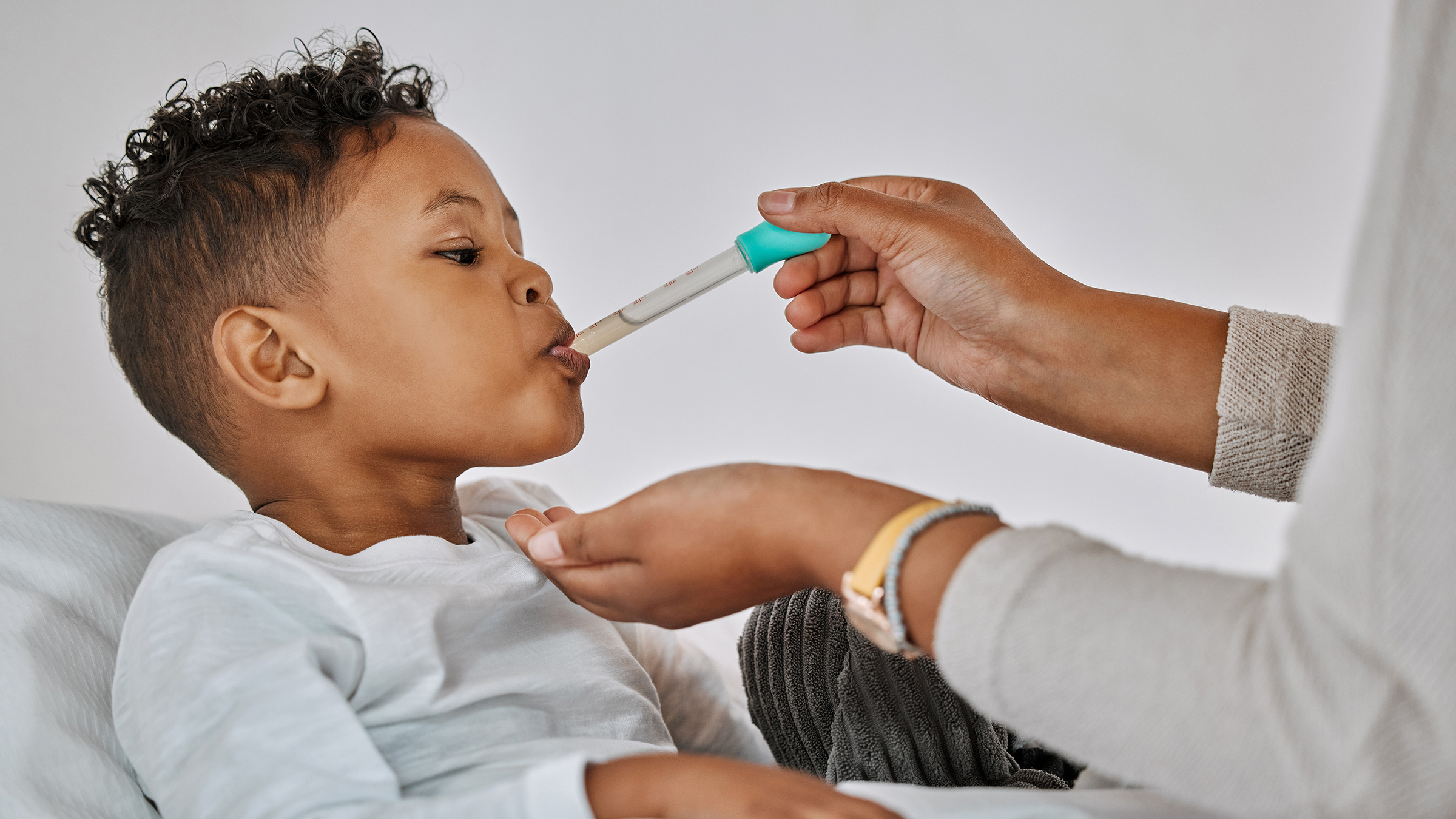 Promethazine Oral Solution, Kofexmalin Baby Cough Syrup, Makoff Baby Cough Syrup and Magrip N Cold Syrup manufactured by Maiden Pharmaceuticals have been flagged by Sahpra. (Photo: GoodRx / Wikipedia)
Promethazine Oral Solution, Kofexmalin Baby Cough Syrup, Makoff Baby Cough Syrup and Magrip N Cold Syrup manufactured by Maiden Pharmaceuticals have been flagged by Sahpra. (Photo: GoodRx / Wikipedia)