Business Maverick
Pfizer submits Covid shot for kids under 5 for FDA authorisation
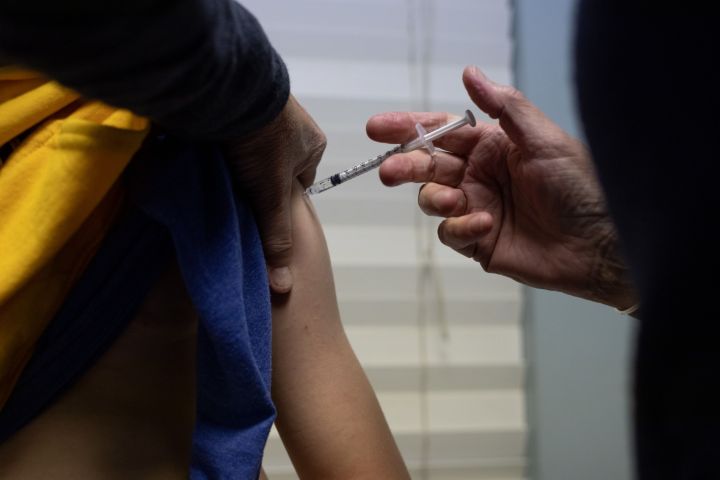
The pharmaceutical company has asked US regulators to clear its Covid-19 vaccine for emergency use in children under age 5, an effort to extend protection against the virus to the country’s youngest.
Pfizer and BioNTech SE finalised their rolling application to the Food and Drug Administration for emergency-use authorization of their vaccine in kids ages 6 months through to 4 years old, the companies said in a statement on Wednesday. The vaccine partners began the submission process in February.
Pfizer and BioNTech announced in late May that a three-shot regimen was highly effective and prompted a strong immune response in children under age 5, based on early results from a highly anticipated trial that is likely to pave the way for infants and toddlers to get immunised.
Read More: Pfizer Covid Shot Seems Protective for Children Under Five
The preliminary analysis found that the vaccine was 80.3% effective in preventing Covid infections among young children, with 10 infections occurring among all participants. Final results will be determined once 21 children enrolled in the study have been infected, according to the companies.
In February, Pfizer and BioNTech launched a rolling review of data from the trial of 1,678 children under the age of 5. The three-dose vaccine regimen was well tolerated, with a safety profile similar to that of the placebo, and most side effects were mild or moderate.
The full submission includes data on the product’s safety, efficacy and ability to prompt an immune response. Children under the age of 5 were a given of series of three doses, each 3 micrograms, or 1/10th the amount given to adults.
With the submission now complete, attention will turn to a forthcoming FDA advisory committee meeting on June 15. The panel of experts will convene to discuss the risks and benefits of the Pfizer-BioNTech and Moderna Inc. vaccines in the youngest children, and provide a recommendation to U.S. regulators as to whether or not they should be cleared for emergency use.
Pfizer said the companies also plan to submit the data to the European Medicines Agency and other regulators around the world. BM




















 Become an Insider
Become an Insider
Comments - Please login in order to comment.