Moving targets
On 27 February, during a farm visit in KwaZulu-Natal, Minister of Agriculture John Steenhuisen told farmers that “the Dollvet vaccine comes in from Turkey, 1.5 million”, with the batch of five million Biogenesis Bagó vaccines to follow a fortnight later. “The date that they’ve given us is 15 March,” he said.
The 15 March delivery did not happen. The department has since confirmed it will arrive before the end of March.
Dipeneneng Serage, the Deputy Director-General for Agricultural Production, Biosecurity and Natural Resources, told Daily Maverick the date was always an estimate. He said that given the many moving parts outside the government’s control, timelines often shifted in both directions.
Complicated process
Getting a foreign-manufactured vaccine into South Africa’s national herd is not a single step. It is a chain, and every link takes time, says Serage.
/file/attachments/orphans/DanielleFMDvaccineholdups_879885.jpg)
The five million Biogenesis Bagó doses are caught in what Serage described as step one: strain matching. Because these vaccines are not manufactured in South Africa, they are not registered here. Before entering the national herd, they must be tested and matched to the specific viral strains circulating locally.
While the first million doses received on 21 February covered SAT1 and SAT2 strains, this larger consignment is multivalent and also works for SAT3, requiring additional testing, said spokesperson for the Ministry of Agriculture, Joylene van Wyk. Matching is currently being conducted by the Pirbright Institute, with results and vaccines expected before the end of March.
The Industry Coordination Council (ICC) has said that speed must be the “overriding priority”. Given the scale of the current outbreak and the number of provinces affected, every step that can be shortened should be.
Once strain matching is complete, the vaccine must still clear other regulatory hurdles before it can enter the country, said Serage.
The importer must apply to the South African Health Products Regulatory Authority (Sahpra) for a Section 21 permit under the Medicines and Related Substances Act, required for any vaccine not registered in South Africa, as well as an import permit under the Animal Diseases Act. The Section 21 application must include technical data on the product’s safety, quality and efficacy, evidence of Good Manufacturing Practice compliance, and reasons why a registered South African alternative cannot be used. Sahpra’s Veterinary Medicines Unit then evaluates the submission and can authorise, request more information or deny it.
“We give all support we can to make the vaccine roll-out a success. We believe that the Minister of Agriculture is seriously trying to turn the tide around,” said Dr David Gerber, CEO of Dunevax Biotech. “Where we do have a problem is with the time it takes for Sahpra to approve the Section 21 permits to ensure [a] continuous supply of vaccines. The process seems to be equally long for all vaccines.”
From landing to field
The journey of the 1.5 million Dollvet doses illustrates the system’s pace and pressure. Expected on 28 February, they landed at OR Tambo on 1 March, delayed by restricted airspace over the Gulf States, according to a statement by the Department of Agriculture. They reached Onderstepoort Biological Products’ Pretoria facilities only on 6 March, as confirmed by social media posts by Dunevax. After the week of 9-13 March, Van Wyk confirmed 98% were in the field.
That is nearly two weeks from landing to the majority of farm deployment. “Every unnecessary delay widens the window in which infection can move, animals remain unprotected, and confidence in the roll-out declines,” said Francois Rossouw, CEO of the South African Agri Initiative (Saai). “A system that takes nearly two weeks to move vaccine from arrival to field-level availability is not a system that should be defending exclusive control over the process.”
/file/attachments/orphans/ED_599277_437704.jpg)
The 1.5 million Dollvet doses have now been allocated across all nine provinces, with KwaZulu-Natal receiving the largest share at 560,000 doses, followed by Gauteng at 213,000 and the Free State at 195,000, according to a release by Red Meat Industry Services on 12 March. A further 75,000 doses are held in reserve for emergency response.
A system not built for this
According to the FMD ICC, the legal framework governing FMD was developed more than four decades ago and was not designed for a multiprovince outbreak of this scale. South Africa’s governance structure, where the national government sets the legal framework, but provinces implement it through their own operational systems, adds further complexity. The ICC has proposed a National Implementation Platform to align national planning with provincial execution and industry capacity.
Dr Frikkie Maré, CEO of the Red Meat Producers’ Organisation, agrees that the system was not ready for what arrived. “We feel that the National Department waited too long to decide on the route to take with FMD,” he said. “It was only in December 2025 that the decision was made to vaccinate the whole country and import vaccines from other manufacturers.”
“What we need right now is a steady flow of vaccines into the country. There is still too much uncertainty about the Section 21 permits that Sahpra should approve,” said Maré. “We further need clarity on the final regulations on [how] the process will work from here.”
Dairy producers welcome the fact that the vaccination roll-out has gained momentum in recent weeks. However, “the pace has been uneven, and many farmers have waited longer than expected before their herds could be vaccinated,” said Fanie Ferreira, CEO of the Milk Producers’ Organisation. “Uncertainty around timelines and vaccine availability remains one of the biggest concerns.”
The capacity debate
On 2 March, Saai, with Free State Agriculture and Sakeliga, filed an urgent court application seeking to prevent the agriculture minister from blocking private vaccine procurement and administration alongside the state roll-out. Mediation is under way. If unsuccessful, the matter will be heard on 24 March.
“The state retains oversight and sets the regulations, while the private sector assists with the government’s own stated objective of mass vaccination,” said Rossouw, describing the proposed parallel system as anything but a free-for-all.
The ICC has confirmed that industry operational capacity exists and is ready to be used, and has formally proposed structures through which that capacity could be channelled into the roll-out. Whether those structures can be activated quickly enough to make a meaningful difference remains the central challenge of the weeks ahead.
With a target of vaccinating 80% of the national herd by December, the vaccines are coming. Whether the regulatory and logistical architecture around them can move at the speed the disease demands is the question. DM






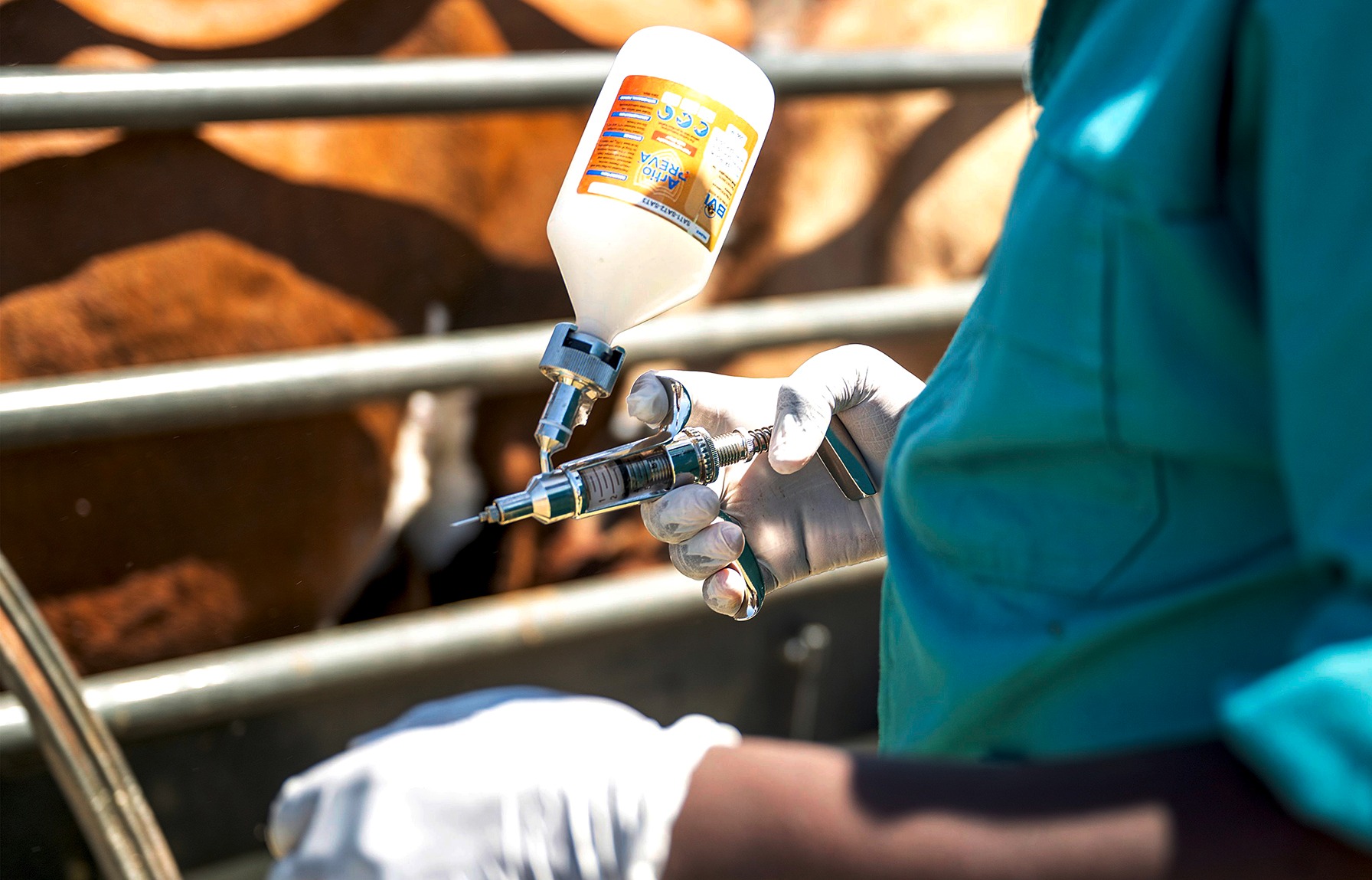 The vaccination process for foot-and-mouth disease in Fisantekraal, Cape Town, on 15 February 2026. The process was aimed at vaccinating cattle in the informal and traditional farming sector. (Photo: Gallo Images / Die Burger / Jaco Marais)
The vaccination process for foot-and-mouth disease in Fisantekraal, Cape Town, on 15 February 2026. The process was aimed at vaccinating cattle in the informal and traditional farming sector. (Photo: Gallo Images / Die Burger / Jaco Marais)