‘The death of a child affects us all. Witnessing the loss of a newborn baby who has sepsis is terribly traumatic, especially so when antibiotics used to treat the child are ineffective,” says neonatologist Professor Sithembiso Velaphi.
“It’s very heavy for a mother to carry her baby, give birth, watch as her newborn gets seriously sick from infection, suffers while being pricked with drips and pumped with drugs to try and save the child – only for her to leave the hospital empty handed. It’s painful,” says Velaphi, who is the head of paediatrics at Chris Hani Baragwanath Academic Hospital in Johannesburg.
/file/dailymaverick/wp-content/uploads/2023/06/Bara-Prof-Sithembiso-Velaphi-pic-byline-Kim-Cloete-for-GARDP.-Feb-2023.jpg.png)
Nurses and doctors feel sad and crushed too when they cannot save a newborn’s life because of antibiotic resistance to bacterial infections. “We need to prioritise the development of antibiotics to treat these babies. For us, success is seeing a baby get better and going home,” he says.
Velaphi was speaking to Spotlight about a landmark global observational study published on 8 June in the journal Plos Medicine, which found that many neonates (within 60 days of birth) get life-threatening bloodstream infections, or sepsis, and are dying because the antibiotics used to treat them are not effective. This is the first global overview to assess the extent of the problem. Spotlight last year reported on interim findings from the same study.
The study, called NeoOBS, led by the Global Antibiotic Research and Development Partnership (GARDP) recruited more than 3,200 babies in 19 hospitals in 11 countries – South Africa, Kenya, Uganda, Thailand, Vietnam, India, Greece, Italy, Bangladesh, Brazil and China.
The researchers reported great variability in mortality rates of babies with sepsis across the 19 hospitals, ranging from 1% to 27.3%.
Sepsis affects up to three million babies a year globally. Importantly, the study’s 80 authors estimate that 214,000 newborns die every year from sepsis that has become antibiotic resistant, and this is mostly in low- to middle-income countries (LMIC). Many survivors suffer from neurodevelopmental problems. Treatment options have become increasingly limited as about 40% of infections are reported to be resistant to standard antibiotic treatments.
/file/dailymaverick/wp-content/uploads/2023/06/VBSADSC_7307-1024x545.jpg.png)
Many infections acquired in hospital
Almost 60% of infection-related deaths were owing to infections acquired at the 19 hospitals under review. Klebsiella pneumoniae was the most common pathogen isolated.
Of the 40 antibiotics approved for use in adults since 2000, only four have included dosing information for neonates in their labelling. Currently, 43 adult antibiotic clinical trials are recruiting patients, compared to only six trials recruiting neonates, researchers say.
New antibiotic treatments are urgently needed, especially in LMICs where almost one in 5 babies with sepsis died. Premature babies are particularly vulnerable to infections because of their immature immune systems.
More than 200 different antibiotic combinations were used by hospitals included in the NeoOBS study, with repeated switching of antibiotics because of high resistance to treatments. This showed a pattern of limited use of the World Health Organisation’s recommended first-line treatment.
Many doctors have had to opt for last-line antibiotics such as carbapenems because of the high degree of antibiotic resistance in their units or because they were the only treatment available.
Outlining various challenges, Velaphi says the risk of infections is very high in hospital settings where there is often a shortage of nurses, beds and space between patients, making it difficult to stop the spread of infection. Chris Hani Baragwanath has an 18-bed intensive care unit (ICU), which is almost always full and when the situation is desperate, there is a spillover of patients into the high-care area. The pressure on the facility is huge and the influx of people from other countries has made it even more challenging, he says.
/file/dailymaverick/wp-content/uploads/2023/06/Neonatal-sepsis-newborn-Pic-byline-Karin-Schermbrucker-for-GARDP-scaled-1.jpeg)
“There is a major problem of infection control, specifically related to high-risk babies – sick babies with complications who need interventions such as drips and even surgery. This increases the chances of infection.
“More than 70% of all deaths ascribed to prematurity at the hospital were due to hospital-acquired multi-drug resistant infections,” he says.
The NeoSep 1 trial
The authors say the NeoOBS study has yielded “a wealth of high-quality data” needed to design trials for much-needed and appropriate treatments for sepsis in babies. Encouragingly, and building on from the observational study, the first global hospital-based neonatal sepsis trial called NeoSep 1 is under way in Kenya and South Africa. Chris Hani Baragwanath is taking part in the trial together with Tygerberg Hospital in Cape Town and the Kenya Medical Research Institute (Kemri) at Kilifi County Hospital. It’s planned that the trial will be expanded to other countries and regions in 2024 with the aim of recruiting 3,000 newborns.
A Personalised Randomised Controlled Trial (PRACTical) design will be used. According to GARDP and partners, the design is a new way of comparing antibiotic treatments for neonatal sepsis. In addition, doctors can choose treatment regimens that are likely to work well for newborns in their specific hospital settings.
Researchers say the development pipeline for new antibiotic treatments is limited and the lack of a universal, effective standard of care creates huge challenges in conducting research to tackle neonatal sepsis. The PRACTical design has been specifically developed to address these challenges in important public health emergencies such as neonatal sepsis. (You can read more about how this type of trial works in the Lancet.)
The trial will compare the safety and efficacy of three new combinations of older antibiotics (fosfomycin-amikacin, flomoxef-amikacin and flomoxef-fosfomycin) against the current standard of care. It will also assess and validate the doses of two antibiotics (fosfomycin and flomoxef) for use in newborns. The trial will also evaluate new combinations of generic antibiotics.
“We are hoping the trial will provide robust evidence that the antibiotic combinations are safe and effective and that this will lead to a change in both WHO and local treatment guidelines,” says Christina Obiero, principal investigator for the NeoSep1 trial for Kemri.
Severity and recovery scores
/file/dailymaverick/wp-content/uploads/2023/06/Neonates-Adrie-Bekker-with-newborn-Tygerberg-Hospital.jpg.png)
The principal investigator for the NeoSep1 trial at Tygerberg Hospital, Professor Adrie Bekker, tells Spotlight: “We have so few antibiotics that work effectively against these very sick babies. And even for those that we have, we are still not 100% sure how to dose these drugs to get accurate concentrations in the blood and to also make sure that the outcomes in these babies are as good as can be. This trial will help give us confidence that we are delivering more effective treatment.”
Bekker, who is also Professor in the Division of Neonatology, Department of Paediatrics and Child Health at Stellenbosch University, says a positive outcome of the NeoOBS study is the development of two important tools which can be used in ICUs globally.
The first is the NeoSep Severity Score, which is a compilation of common symptoms and signs that can occur in a baby with clinical sepsis. The second is the NeoSep Recovery Score, which will assist clinicians in deciding if they can stop antibiotics earlier.
The tools are expected to help prevent the often excessive and inappropriate use of antibiotics for over too long a period, which compounds the problem of antibiotic resistance globally.
Diagnosis in older age groups, children and adults is generally easier.
“It’s sometimes difficult for a clinician to know whether a baby actually has neonatal sepsis because it can present very subtly and not always with the same symptoms,” Bekker explains.
The blood culture is the gold standard for diagnosing neonatal sepsis, but Bekker says only around 10% of blood cultures will grow an organism even if the baby has sepsis, making it very difficult to get a diagnosis. “And because it’s such an aggressive disease and a baby can die very quickly from it, clinicians tend to rather over-treat than under-treat. That is correct but, just as important as it is to start antibiotics quickly, it’s important to stop them if they are not necessary. The NeoSeps Severity score will help doctors identify babies that are at very high risk from sepsis and those that would need treatment immediately.”
Velaphi says a major challenge is the time it takes for an outcome of the blood culture and the general protocol is to start antibiotics immediately. Waiting between 24 to 48 hours can be too late for a child who may have sepsis and could die. On the other hand, antibiotics may be given to children who do not have sepsis and this adds to the frequency of antibiotic resistance. “So, you are damned if you do and you are damned if you don’t.”
He says we need new diagnostic tests that are reliable and have a high degree of sensitivity and specificity. “We need antibiotics that work to reduce mortality,” he adds. DM
This article was published by Spotlight – health journalism in the public interest.






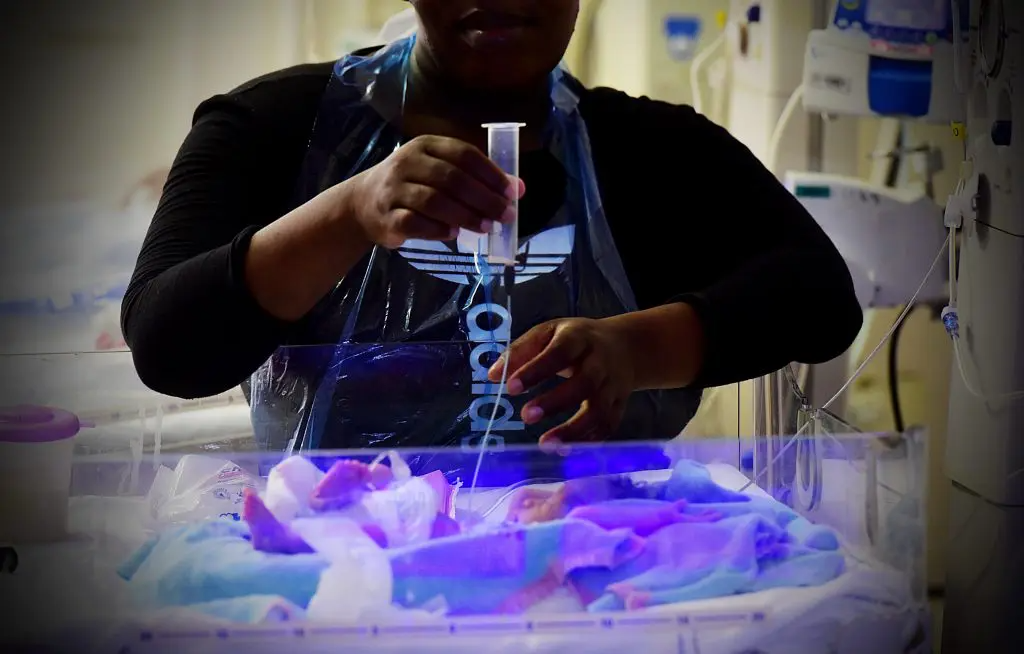 Almost 60% of infection-related newborn deaths were due to infections acquired at the 19 hospitals under review. PHOTO: Denvor de Wee/Spotlight
Almost 60% of infection-related newborn deaths were due to infections acquired at the 19 hospitals under review. PHOTO: Denvor de Wee/Spotlight