The number of newborn babies dying from neonatal sepsis is rising as the antibiotics used to treat them are not working effectively, a landmark international study has found. Neonatal sepsis is a life-threatening bloodstream infection that affects up to three million babies a year globally.
The Global Antibiotic Research and Development Partnership (GARDP) and partners recently released their findings showing that antibiotic resistance to bacterial infections, and the increased use of broader-spectrum antibiotics have led to high levels of mortality and neurodevelopmental problems in surviving babies.
Babies are especially vulnerable to infections because of their underdeveloped immune systems, says Sally Ellis, Children’s Antibiotics Project Leader for GARDP. “Compounding the problem is that babies are dying because of the lack of good treatment options."
The observational study, called NeoOBS, of more than 3,200 newborns with neonatal sepsis was conducted at 19 hospitals in 11 high-, middle- and low-income countries, including South Africa. It assessed antibiotics currently used to treat newborns with sepsis, and the extent to which resistance makes these treatments ineffective.
Often deadly
The three-year study across four continents found overall mortality of 11% at 28 days in babies suspected of having neonatal sepsis. The rate jumped to 18% in cases where a diagnosis was confirmed by culturing the pathogen from blood samples.
More than half of infection-related deaths (59%) were due to hospital-acquired infections. Klebsiella pneumoniae was the most common pathogen isolated and is most often associated with hospital-acquired infections, which are increasingly resistant to existing antibiotic treatments, according to a GARDP report accompanying the results.
A concerning trend identified was the frequent prescribing of last-line antibiotics such as carbapenems because of the high degree of antibiotic resistance in health facilities. Of note, 15% of babies with neonatal sepsis were given last-line antibiotics. Other key findings are that in-hospital mortality was high but variable ranging from 1% to 27% across sites.
Antibiotic prescribing practice varied markedly with limited use of the World Health Organization’s recommended regiments in many hospitals due to high antibiotic resistance. GARDP has identified potential candidates to replace the WHOs’ recommended first-line treatment.
‘Systemic failure’
According to the GARDP report, policymakers, governments, institutions, and companies have for decades neglected to address collectively a “systemic failure that is leading to the avoidable deaths of babies from neonatal sepsis, especially in low-income countries”.
Principal Investigator Mike Sharland from St George’s, University of London said, “There are virtually no studies underway on developing novel antibiotic treatments for babies with sepsis caused by multidrug-resistant infections. This is a major problem for babies in all countries, both rich and poor.”
He says the study has enabled GARDP to design a new global trial of new treatments to reduce mortality from neonatal sepsis. The NeoSep1 trial will begin in South Africa and Kenya later this year, with up to eight more countries participating in 2023. It will test, among other objectives, the safety and effectiveness of three potential new antibiotic combination treatments, ranking them against existing commonly used antibiotic regimens.
A total of 600 babies of the 3,200 babies in the NeoOBS study were from three South African hospitals, namely Charlotte Maxeke in Johannesburg, Chris Hani Baragwanath in Soweto, and Tygerberg Hospital in Cape Town. All three tertiary hospitals represent a mix of newborn babies with medical problems including prematurity, and babies needing surgery including for congenital abnormalities.
Speaking to Spotlight, Professor Angela Dramowski, principal investigator and Head of the Clinical Unit: General Paediatrics at Tygerberg Hospital says globally neonatal survival is “crucial”. The latest data is that half of all deaths for children under five years are in the first 28 days of life. “If we want to improve child survival we have to focus on neonatal survival,” she says. “A baby born today in an African country has a ten-fold risk of mortality compared to a similar-sized baby born in a high-income country.”
While there has been a huge improvement in child survival, neonatal survival has stagnated in most African countries, to the point where 95% of all newborn deaths globally happen in low- to middle-income countries, at least half to two-thirds in Sub-Saharan Africa, she says.
The big three killers
Reasons for deaths, she says, include the vulnerable neonatal period in particular, with the “big three killers” being prematurity, lack of oxygen, injury at birth, and infection.
She says Infection is often thought to be the least prevalent, but a study at Chris Hani Baragwanath Hospital in 2019 showed as many as 74% of all the deaths currently ascribed to prematurity could be linked back to severe hospital-acquired multi-drug resistant bacterial infections.
Dramowski says South African data in the Saving Babies Reports and the Perinatal Problem Identification System attribute about 10% of neonatal deaths to sepsis but, in reality, she says it’s likely to be far higher and will increase in future.
Factors that will add to the neonatal sepsis burden in Africa include huge population growth and rapid urbanisation. “This means more in-hospital delivery of babies and huge growth in the public sector maternity and neonatal patient care load. This translates to overcrowding, and even lower staff to patient ratios for a very vulnerable population group.”
Lack of infection control is another problem. “It’s very difficult to pay attention to the basics which make health care safe. In a busy neonatal unit with a huge turnover of babies, simple hand hygiene, cleaning up the environment, cleaning of shared equipment are not, unfortunately, optimally applied in most South African settings,” she says.
In the Western Cape neonatal units, she says, in the nine big hospitals across the metro and beyond, occupancy rates are more than 90%. In the Eastern Cape, many of the hospitals are at 140% to 150% capacity daily. “So, you can’t even ensure adequate spacing between individual babies and mothers — a recipe for a very easy transmission of bacteria.
“We are getting very resistant bacteria that are difficult to treat so we are increasingly using antibiotics of last resort, meaning antibiotics that are not fully effective, resulting in the chance of more babies dying or surviving with a disability,” she says.
In addition, she says, “We have to go back to using drugs that were abandoned in the past because they were too toxic, not licenced for babies, for example, Colistin, which is a very old antibiotic that had to be revived to treat neonatal sepsis caused by Acinetobacter. This a huge problem now, particularly at the two Johannesburg sites where they had Acinetobacter, the most common pathogen that they cultured from babies with sepsis.”
When these antibiotics are ruled out, Dramowski says neonatologists and paediatricians opt for the “big gun” — a broad-spectrum antibiotic called meropenem which is in the carbapenem class of antibiotics. “But in the last few years, we’ve had some big outbreaks at several hospitals in South Africa of carbapenem-resistant enterobacterales, a group of gut-based organisms. One of these is Klebsiella which is the most common and is causing between 5% and 10% of all infections. “We are using these last-line antibiotics at our hospital at Tygerberg in up to two-thirds of babies with sepsis because we can't afford to get it wrong. So, we often go in with our most powerful antibiotics upfront to try to save the baby and then try to de-escalate our antibiotics to target the specific bug that we culture.”
Diagnostics and detection
Dramowski says globally tests are very limited, not very sensitive or specific, making it difficult to distinguish whether a baby has deteriorated due to causes related to prematurity or due to infection.
“Across Africa where there are few microbiology laboratories and diagnostic tests, units will opt to give very broad spectrum, very powerful antibiotics upfront, because they want to save lives and they are not sure whether this is an infection or not. And the spin-off of that is more babies get treated with antibiotics more often, which removes the good bacteria from the baby’s gut and replaces them with bacteria that are antibiotic-resistant so the next time the baby gets an infection you need to give it even more powerful, more broad-spectrum antibiotics to treat the next infection. So, you really are treating blindfolded or in the dark. And that’s why the new NeoSep1 study is going to be hugely helpful because we’ll get an idea of which bacteria resistance profile and which antibiotic regimens had the best treatment outcomes across a wide range of settings,” she says.
Dramowski says It can take up to three to four days to identify the bacteria and the sensitivity of the bacteria to the antibiotic by which stage many babies will have died waiting for their results. In addition, in many settings, the antibiotics needed to treat infections are not even available at most hospitals.
The NeoOBS study represents settings where “you have probably the worst of infection but also the best access to a broad range of antibiotics and ICU facilities”. “So, the mortality estimates to me look quite conservative where they had about 18% mortality. But, if you are working in a smaller district hospital with no access to ICU, the odds of a baby with a very serious bacterial infection dying may be as high as 50% or more.”
Dramowski says better diagnostics and better drugs are needed and the “holy grail” is to prevent these infections from happening in the first place.
“South Africa needs to do better. We know we have recurrent outbreaks in neonatal units, understaffing, and overcrowding. We need to allocate resources to physical infrastructure to make neonatal and maternity care safer, [and] a skilled workforce that understands infection control, and national surveillance for infections in hospitalised neonates. This will help to identify hotspots and will reduce the [number] of antibiotics needed to treat infections that are often preventable," she says. DM/MC
*This article was published by Spotlight – health journalism in the public interest.

[hearken id="daily-maverick/9472"]





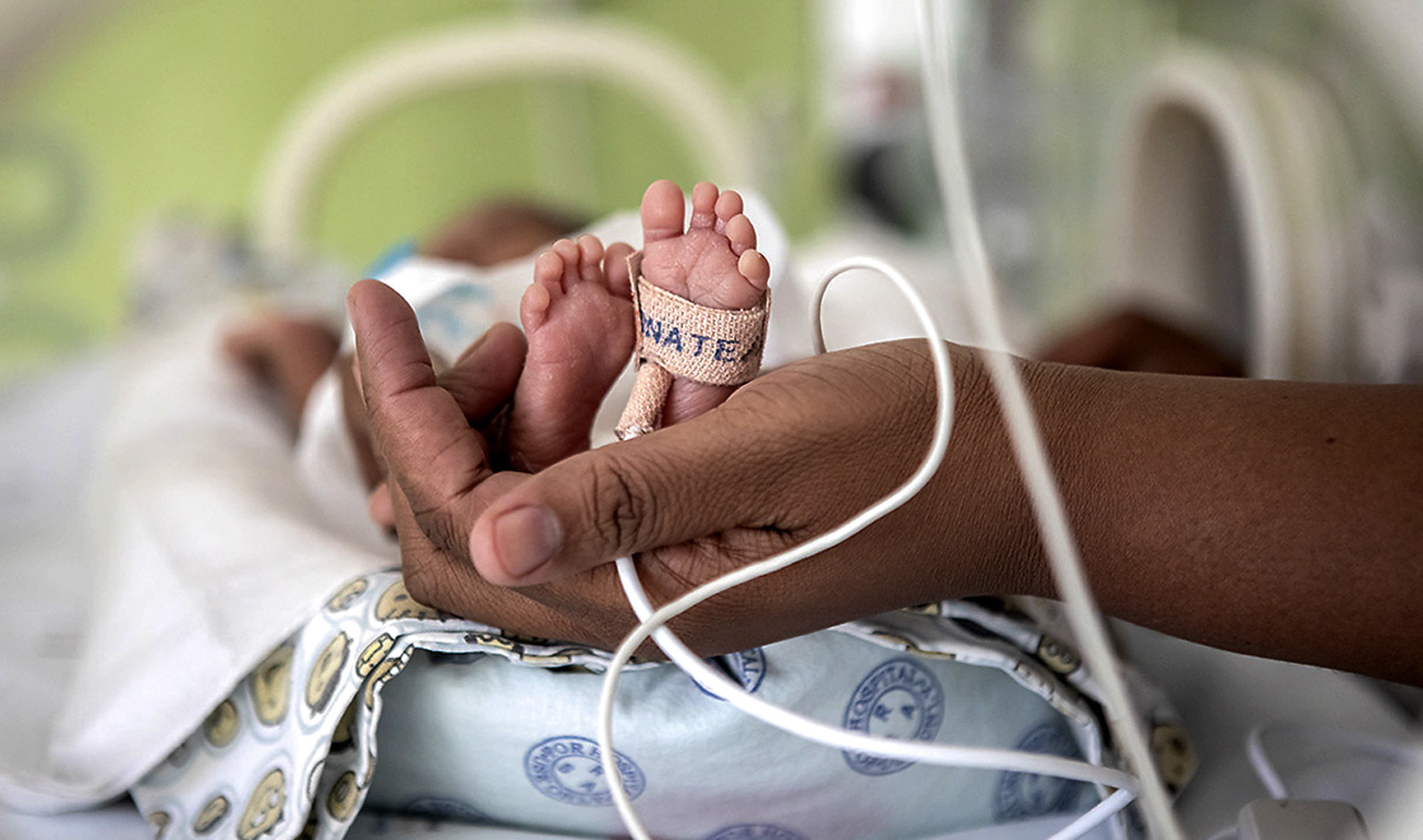 Newborns at Charlotte Maxeke Johannesburg Academic hospital (Photo: Karin Schermbrucker, for GARDP)
Newborns at Charlotte Maxeke Johannesburg Academic hospital (Photo: Karin Schermbrucker, for GARDP)