Silence
Phumeza Tisile was barely out of her teens when she woke up one morning, turned on the television and found there was no sound. It became alarming when she went to the bathroom and couldn’t hear the running water either. Maybe she just had something in her ear, she told herself. Just stay calm; you’re going back to the doctor.
Tisile had been on a weekend pass out of Brooklyn Chest Hospital, Maitland. It was 2010; the excitement over hosting the Soccer World Cup had reached fever pitch. Tisile was watching from the sidelines – she was being treated for multi-drug resistant tuberculosis (MDR-TB).
“I went back to Brooklyn Chest Hospital,” recalls Tisile. “The next morning I told the nurse that I couldn’t hear. I saw her moving her lips saying something but I couldn’t make out what she was saying. She wrote down that I had to go to the audiology department, where I did some tests. The audiologist wrote down that I was deaf. That was the most confusing thing ever. He said I had to go and speak to my doctor. I asked the doctor how this was possible. She wrote that it was the side effect of the Kanamycin injection and there was nothing they could do about it.
“No one had told me about the side effects of the drugs.”
Tisile was moved to another ward with four other patients who were also deaf from the same medication. At least she wasn’t alone, she thought. “I had plenty of music in my phone, which was useless now. I decided to delete them all, and gave away the headphones. I really tried to convince myself that it was no big deal; that it would pass.” But it did not pass, and it was a big deal.
“Then it got scary. Two of the patients committed suicide, the other one ran away from the hospital.”
Five years later, Tisile got a cochlear implant. “Tears of joy,” she says, recalling the moment she heard her mother’s voice for the first time. “There were tears, but they were tears of joy.”
Today, Tisile is a passionate advocate for TB patients. Broadly, she says, she is advocating for change around TB medication. She co-authored the Drug-Resistant TB Manifesto, presented to world health leaders in Geneva.
Tangle
Tisile is fighting alongside her doctor, Dr Jennifer Hughes. Tisile is one of the lucky ones; she survived the disease. Many do not. The impending arrival of delamanid, a new drug said to achieve excellent outcomes for MDR-TB patients – especially those with HIV – was announced with great fanfare to South Africans earlier in 2016.
The National Department of Health was to negotiate with Japanese manufacturer Otsuka to roll out a clinical access programme – a pilot project which would make the drug available to a small number of patients who had not responded to the more widely used TB treatments.
But hopes have stalled, tied up in a tangle of red tape and debates over pricing.
In a nutshell, delamanid is one of only two new TB drugs – the other being bedaquiline, for which a clinical access programme was rolled out a few years ago – to have been developed in the last 50 years. South Africa was, in fact, one of the sites of its clinical trials. It could potentially change the prognosis for thousands of patients in South Africa.
It dramatically changes the picture for those with drug-resistant TB, according to MSF and the World Health Organisation (WHO)’s Global TB Report.
But approaching the end of 2016, the drug – which has been approved for use in a number of countries not facing a significant TB threat – has still not been registered in South Africa.
“Otsuka’s delays in registering delamanid with the Medicines Control Council are limiting treatment to a select few,” MSF’s spokesperson Angela Makamure said in a statement on Wednesday. “South Africa has one of the highest burdens of TB and DR-TB in the world, with more than 20,000 people diagnosed with DR-TB in 2015. Yet the current DR-TB treatment is only successful in about half of all people who receive it.”
Hughes says delamanid is of particular interest in South Africa because it can be taken with standard, fixed-dose combination treatment for HIV, and over 70% of people diagnosed with TB are also HIV-positive.
“An estimated 7,000 DR-TB patients per year in South Africa could benefit from the inclusion of delamanid in their treatment regimens, but we need Otsuka to act rapidly to register the drug and provide an interim access plan if we want people who need this drug to receive it,” she says.
Registration would allow delamanid to be incorporated more readily into treatment guidelines and purchased on national tender, all of which would improve availability of the drug for patients with DR-TB.
First irony: South Africa has the potential to be a trailblazer in TB drug distribution, thanks to a proactive approach by the NDOH. “MSF is treating some of the lucky few people in South Africa — and in fact the whole world – to receive access to strengthened treatment regimens including bedaquiline and delamanid,” says Hughes. “South Africa’s Department of Health has been a leader in accessing new DR-TB drugs, with over 1,750 patients receiving bedaquiline nationally since 2013. [But] despite progress, all DR-TB patients are likely to benefit from better treatment regimens, and access to new drugs on such a scale is nowhere close to a reality.”
In contrast to bedaquiline, though, only a handful of DR-TB patients in South Africa have had delamanid included in their treatment by having their clinicians apply on a case-by-case basis for compassionate use. This process is time-consuming and by no means guaranteed.
MSF has also purchased the drug – at an eye-watering R23,600 per six-month treatment course – for five beneficiaries in Khayelitsha. The Khayelitsha patient cohort is the largest in South Africa.
MSF says the clinical access programme for delamanid, while welcome, has also faced its share of timing challenges. “The eventual start of a clinical access programme nationwide will be a welcome development,” Hughes says, “though it is unclear if there will be a sufficient number of treatment courses to meet demand for delamanid prior to local registration of the drug.”
Second irony: this week, delegates from 120 countries are meeting at the Union World Conference on Lung Health. But TB has overtaken HIV/Aids as a killer worldwide. And South Africa is not the only country struggling to access the new life-saving drugs.
Otsuka says it is playing ball; it has donated 400 delamanid treatments previously and has also reduced its prices for poorer countries. The trouble is that – even if the administrative delays are sorted out, even if the discounts are given – access isn’t cheap.
Otsuka is working with the WHO-led Stop TB Partnership’s Global Drug Facility to help more than 100 low- and middle-income countries gain access to delamanid. Under the new partnership, countries in South-East Asia and the Pacific can buy delamanid for a 95% reduction in price. But the reduced price is still $1,700 for a course.
The way out
Sixteen-year-old Sinethemba Kusa was one of the first South Africans to access delamanid. She fell ill soon after losing her aunt, who raised her after her mother died. Her grandmother, now her caregiver, has appealed to Otsuka to apply for registration to avoid further delays.
“TB is a giant but not a killer,” she says.
But with enough administrative and financial hurdles, it can be. The NDOH has proactively taken steps to establish the clinical access programme with Otsuka, it says. But with Otsuka dragging its feet on registration, its hands are tied.
There is one thing that can be done locally, however: re-examining patent laws. Earlier this year, the Fix the Patent Laws campaign, which includes TAC and Section27, demonstrated against legislation that ultimately benefited pharmaceutical companies and not patients. It is fighting for all governments to urgently adopt and act on UN recommendations to safeguard the right to health, and urged South Africa’s Department of Trade and Industry (DTI) to lead the way for BRICS.
This can have a major impact on drug prices. According to MSF, a recent study of the production costs for new DR-TB drugs has shown that prices could come down by 95% if robust generic competition were introduced — even after allowing for a reasonable profit margin.
“Currently people are suffering and dying because medicines for cancer, hepatitis, tuberculosis, mental health and many other diseases are too expensive,” said Nkhensani Mavasa, National Chairperson of the Treatment Action Campaign, after some 1,000 activists supporting the Fix the Patent Laws campaign marched to the DTI in Pretoria in September. “While we won the fight for first line HIV medicines, we lost the battle for almost all other medicines.”
The campaign is determined to drive the DTI to re-examine its commitment to reforming South Africa’s patent laws in a draft National Policy on Intellectual Property published in 2013, and its application to medicine – and also to consider the UN Secretary General’s High-Level Panel Report on Access to Medicines.
All of the above, however, is likely to take time – time patients don’t always have.
In the meantime, says MSF, it is urgent for Otsuka to file its registration. “Otsuka has yet to file for registration of delamanid in South Africa, a process which can take years, even when applications are expedited,” the organisation said.
As for the patients, they simply wait and see if they will be lucky. “Thank you to the people who invented delamanid, because it can change a lot of lives,” Sinethemba wrote on her blog detailing her recovery. May there be many more blogs like hers. DM
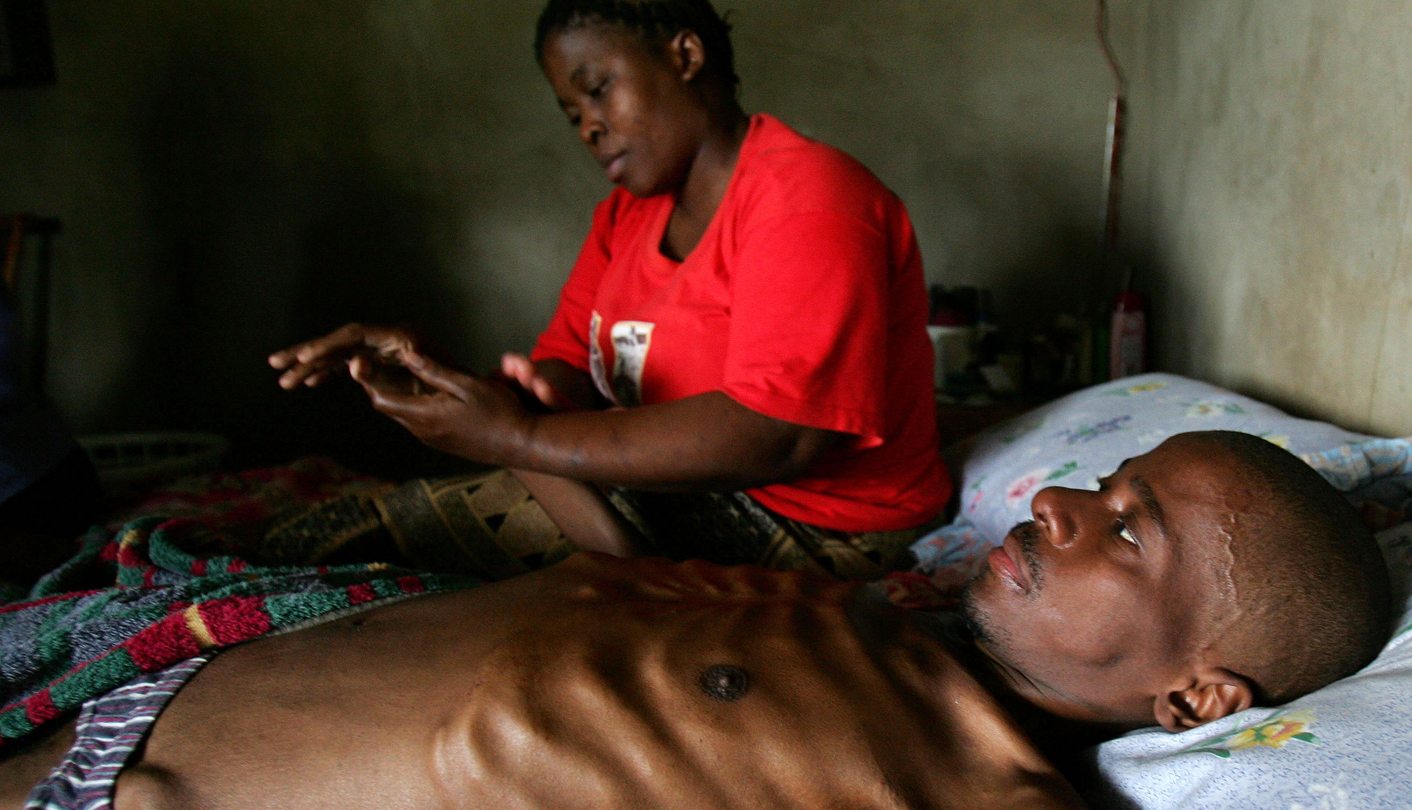
Photo: Hlengewi Mthembu massages the hands of her husband Nkosinathi at their home in Esikhawini township, near Empangani, in South Africa’s KwaZulu-Natal province October 13, 2005. REUTERS/Mike Hutchings